Transmission from Ricardo
📣 Before We Dive In
💬 Join the Discord! Weekly signal deserves weekly debate. Our community is where readers pressure-test the Big Picture takes, share primary sources, and flag stories before they hit the newsletter. Join here
🗣️ Tell me what's working! This newsletter improves when you push back. Two minutes, six questions: respond here. Every response gets read.
🤝 Check out Mintlify
Table of Contents
🧠 Weekly Signal
Neurotechnology News | April 4–10, 2026
Let's cut through the noise.
This week, researchers in Colorado pushed BCI implants into higher-order cortical territory while long-term DBS outcome data clarified target-selection tradeoffs for young-onset Parkinson's. Meanwhile, CorTec became the first company worldwide to earn FDA Breakthrough Designation for a stroke-rehab BCI. On the policy side, Nature called for brain organoid governance just as DARPA opened proposals to turn those same organoids into drone computers. Bottom line: the field is racing to write back to the brain faster than anyone is writing rules for it.
⚡ Signal of the Week
CorTec's FDA Breakthrough Device Designation for the Brain Interchange system is the week's defining story. The designation validates a fundamentally different BCI architecture than the read-only systems dominating headlines: CorTec's 32-channel, fully wireless, closed-loop implant both records cortical activity and delivers targeted stimulation back in real time. It's also the first BCI ever designated for stroke motor rehabilitation — an indication that moves the field from a communication-assistance niche serving dozens of patients toward a therapeutic market addressing millions of stroke survivors. From an engineering standpoint, closing the loop introduces real-time control problems — latency budgets, feedback stability, stimulation artifacts — that read-only BCIs have never had to solve. If CorTec's IDE trial holds up, every BCI company will have to answer a strategic question: are you building a sensor, or a controller?
🔬 SCIENCE & CLINICAL TRIALS
🟢 Colorado team implants BCI in higher-order cortex
Published: April 9, 2026
Read more ➡️ Mirage News / CU Anschutz Medical Campus announcement
Neurosurgeons at UCHealth University of Colorado Hospital implanted Blackrock Neurotech Utah Arrays into a 41-year-old man paralyzed for a decade, targeting higher-order cortical regions involved in planning and decision-making rather than primary motor cortex alone. The Caltech-led consortium (PI: Richard Andersen) also plans sensory cortex stimulation to restore touch.
💡 Big Picture: Moving upstream in the motor pipeline trades signal clarity for richer semantic content — decoding intent instead of execution redefines where in the neural stack a BCI should tap.
🟢 Long-term DBS data clarifies STN vs. GPi tradeoffs
Published: April 6, 2026
Read more ➡️ UF Fixel Institute
A University of Florida retrospective study found that subthalamic nucleus (STN) deep brain stimulation (DBS) produced greater motor-score improvement and medication reduction at 5 years in young-onset Parkinson's patients, while globus pallidus interna (GPi) stimulation better controlled dyskinesia and motor fluctuations. The single-center, nonrandomized design introduces selection bias — target choice remained clinician-driven, not algorithmic.
💡 Big Picture: Closed-loop DBS controllers need an objective function, and this data exposes how far the field remains from biomarker-driven target selection versus clinical intuition.
💼 INDUSTRY & STARTUPS
🟢 CorTec earns FDA Breakthrough Designation for closed-loop stroke BCI
Published: April 8, 2026
Read more ➡️ MassDevice / GlobeNewswire
CorTec GmbH became the first company globally to receive FDA Breakthrough Device Designation for a BCI targeting chronic stroke motor rehabilitation. The Brain Interchange system uses 32 dual-purpose record-and-stimulate channels, ceramic encapsulation for long-term hermeticity, and demonstrated 500-day signal stability in a preclinical ovine study published in Nature Scientific Data; the first human patient at the University of Washington (implanted July 2025) has shown upper-limb motor recovery beyond the conventional-therapy plateau.
💡 Big Picture: Fewer channels doing double duty (read + write) is an architectural bet against the electrode-count arms race — CorTec optimizes for closed-loop therapeutic bandwidth, not raw decoding throughput.
🏛️ POLICY & ETHICS
🟢 Nature calls for international oversight of brain organoids
Published: April 8, 2026
Read more ➡️ Nature News Feature / Nature editorial
Nature's editorial board argued that multi-region assembloids — now capable of driving muscle contraction and modeling four-part sensory-to-cortex pain processing — have reached an inflection point requiring a dedicated, sustained international oversight entity. The editorial opposes a moratorium but notes researchers hope to launch the first clinical trial of an organoid-developed treatment this year.
💡 Big Picture: Current frameworks (FDA for devices, IRBs for human subjects) have no classification for self-organizing biological circuits that compute — a governance gap that only widens as organoids move from research tool to engineering substrate.
🟢 DARPA opens proposals for biological processing units
Published: April 10, 2026 (Proposers' Workshop)
Read more ➡️ Defence Blog / DARPA O-Circuit program page
DARPA's Biological Technologies Office held the Proposers' Workshop for O-Circuit, a 42-month program to develop biological processing units (BPUs) from living neural, glial, and immune cells. Task Area 1 benchmarks BPUs against game environments (Ms. Pac-Man); Task Area 2 integrates them with olfactory receptor neurons on drone platforms to detect explosives. Proposals are due May 11; Phase 1 begins November 2026.
💡 Big Picture: A fly brain navigates on ~6 mWh/day — orders of magnitude below any silicon equivalent — and DARPA is betting that power efficiency justifies operating on a substrate that is inherently alive and mortal.
🔍 Open Question This Week
The same week Nature demanded sustained international oversight of brain organoids, DARPA opened proposals to deploy those organoids as inference engines on military drones. The frameworks being discussed assume organoids are scientific tools; DARPA's program treats them as engineering components. No existing regime — FDA for devices, IRBs for human subjects, ITAR for defense exports — governs a living biological neural network used as a compute substrate in the field. Where does neurons-in-a-box regulation live, and who owns writing it?
🤝 This week’s partnership
We killed RAG and sandboxes. Here's what we built instead.
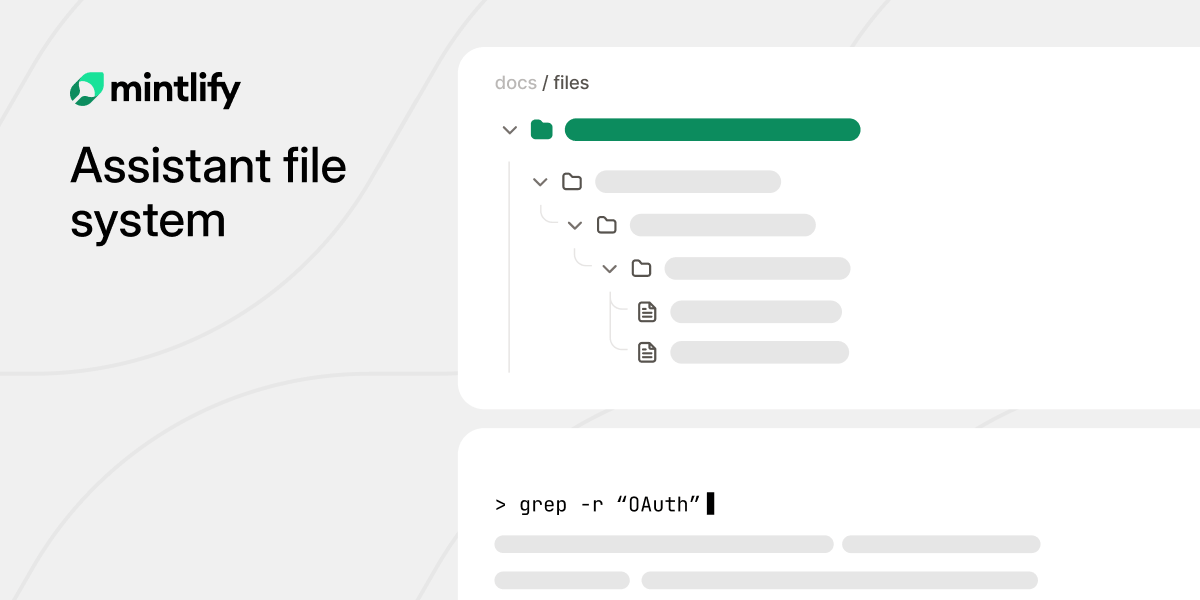
Our documentation assistant had a problem. RAG pipelines only sent the model page fragments, making responses feel half-baked. The fix was obvious: give the agent full doc access. Quality jumped, but startup hit 46 seconds and costs ballooned to $70K+/year.
The insight: the agent doesn't need a real filesystem. It just needs to think it's in one.
We built ChromaFs, a virtual filesystem that translates standard UNIX commands into queries against our Chroma database. Every doc page becomes a file, every section a directory. The agent explores documentation the way a developer explores a codebase.
The results: startup dropped from 46s to ~100ms, marginal compute cost hit $0 per conversation, and output quality matched the full sandbox, with access control built in.
ChromaFs now powers 30,000+ daily conversations for hundreds of thousands of users. No containers, no cold starts, no invoice surprises.
Mintlify powers docs for 20,000+ companies, reaching 100M+ people a year. We just raised a $45M Series B led by a16z and Salesforce Ventures to keep building the knowledge layer for the agent era.